|
8/21/2023 0 Comments Types of crystal lattice 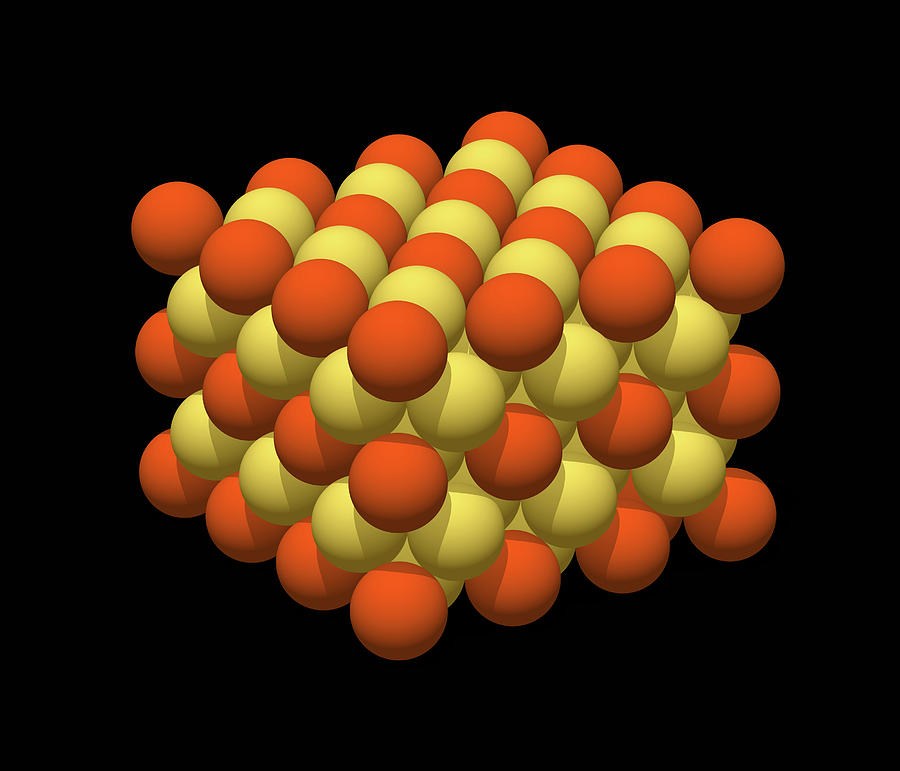
These patterns are located upon the points of a lattice. In a crystal structure, there is a pattern of arranging the atoms or units.When a group of molecules tends to arrange each unit repeatedly at a lattice point, a crystal is made. Lattice describes the structure of crystals.What is the difference between Lattice and Crystal? Crystals have an aesthetic value, and it is believed to have healing properties thus, people use them to make jewelry. Crystals can have different shapes and colors. sodium chloride) and molecular crystals (e.g. Crystals can be categorized according to their physical and chemical properties. There are water-based crystals in the form of snow, ice or glaciers. For example, calcite is produced by mollusks. Crystals are formed by living organisms too. Crystals are naturally occurring on earth as large crystalline rocks, such as quartz, granite. The atoms, molecules, or ions in crystals are arranged in a particular manner, thus have a long-range order. Both of these covalent lattices have a very high melting point, and they cannot conduct electricity.Ĭrystals are solids, which have ordered structures and symmetry. (Diamond is known to be one of the strongest minerals.) Quartz or silicon dioxide also has covalent bonds, but they are between silicon and oxygen atoms (lattice of different atoms). Diamond, by forming a structure like this, has gained a high stability. Therefore, each carbon atom has the tetrahedral arrangement. 
Diamond is composed of only carbon atoms, and each carbon atom is covalently bonded to four other carbon atoms to form the lattice structure. Normally lattice enthalpy is exothermic.ĭiamond and quartz are two examples for three-dimensional covalent lattices. Lattice energy or enthalpy of lattice is the measure of the strength of the ionic bonds in the lattice. The number of ions present in the lattice varies with the size of it. Due to all the attractions between ions, the lattice structure is more stable. In the lattice, each sodium ion is surrounded by six chloride ions, and each chloride ion is surrounded by six sodium ions. Chlorine is a nonmetal and has the ability to form a -1 charged anion. Sodium is a group 1 metal, thus forms a +1 charged cation. 
For example, sodium chloride can be taken. When ions joined with ionic bonds, they form ionic crystals. 
Lattices are crystalline structures with these repeated basic units. The basic unit can be an atom, molecule or an ion. It can be defined as a solid, which has a three- dimensional ordered arrangement of basic units. In chemistry, we can see different types of ionic and covalent lattices. These two words are interchangeably used, but there is a small difference between the two. Rock candy, the crystalline form of table sugar or sucrose, is an example of a molecular crystal.Lattice and crystal are two words that go hand in hand. Molecular crystals tend to be soft with relatively low melting points. A molecular crystal is held together by non-covalent interactions, like van der Waals forces or hydrogen bonding. Molecular Crystals: These crystals contain recognizable molecules within their structures.Table salt (NaCl) is an example of this type of crystal. Ionic crystals are hard and have relatively high melting points. Ionic Crystals: The atoms of ionic crystals are held together by electrostatic forces (ionic bonds).Metallic crystals tend to be very dense and have high melting points. This leaves the outer electrons of these atoms free to float around the lattice. Metallic Crystals: Individual metal atoms of metallic crystals sit on lattice sites.Examples of covalent crystals include diamond and zinc sulfide crystals. Many covalent crystals have extremely high melting points. You can think of a covalent crystal as one big molecule. Covalent Crystals: A covalent crystal has true covalent bonds between all of the atoms in the crystal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
